
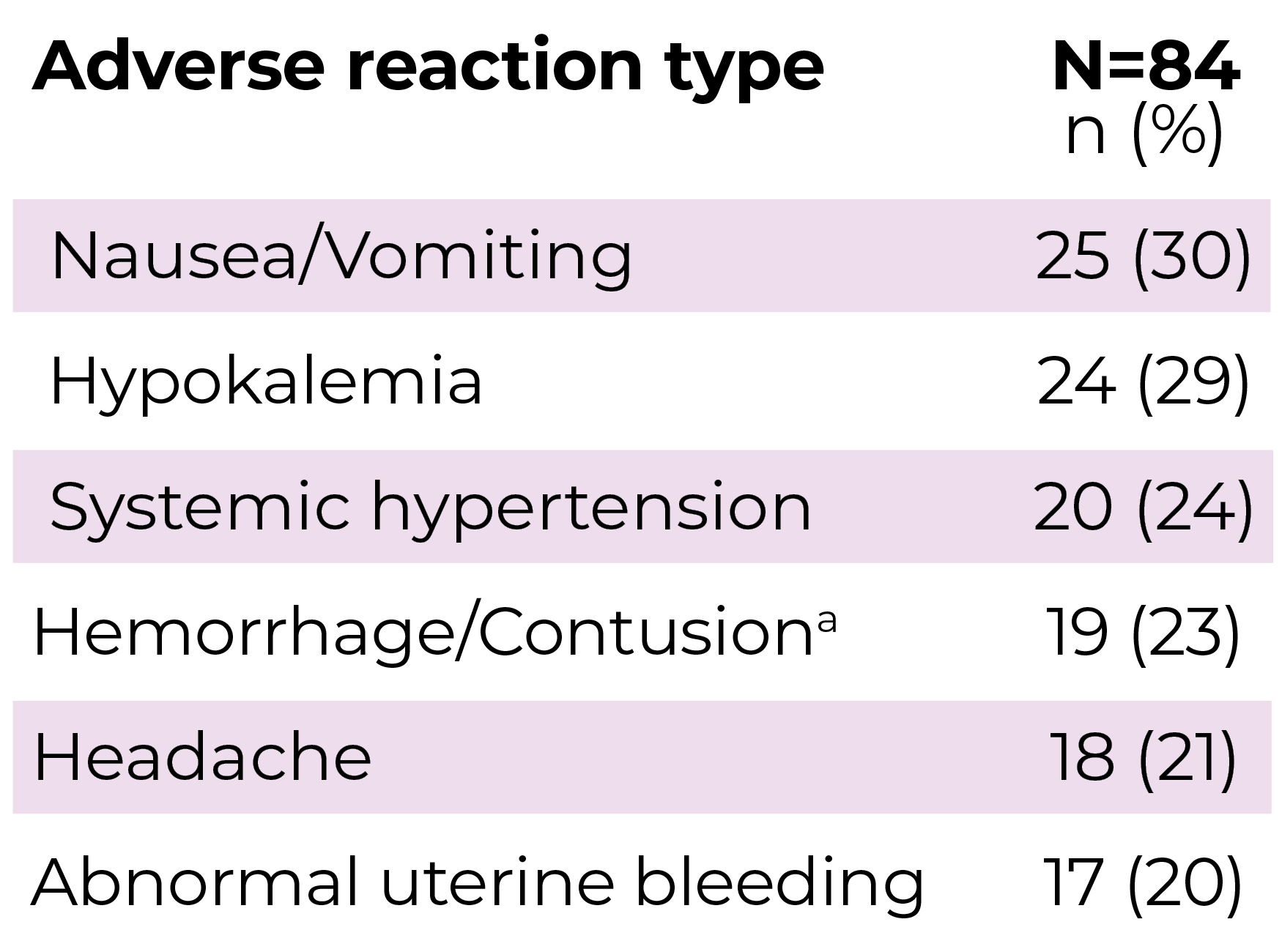
aHemorrhage/contusion includes blood urine present, epistaxis, eye hemorrhage, gingival bleeding, hematoma, hematuria, hemorrhoidal hemorrhage, melena, and scleral hemorrhage.
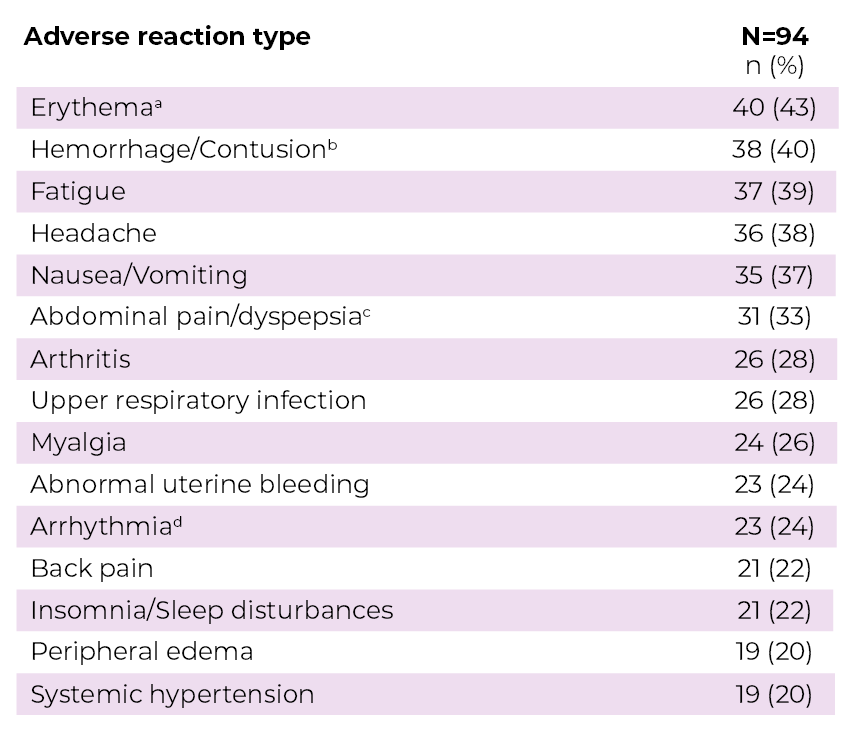
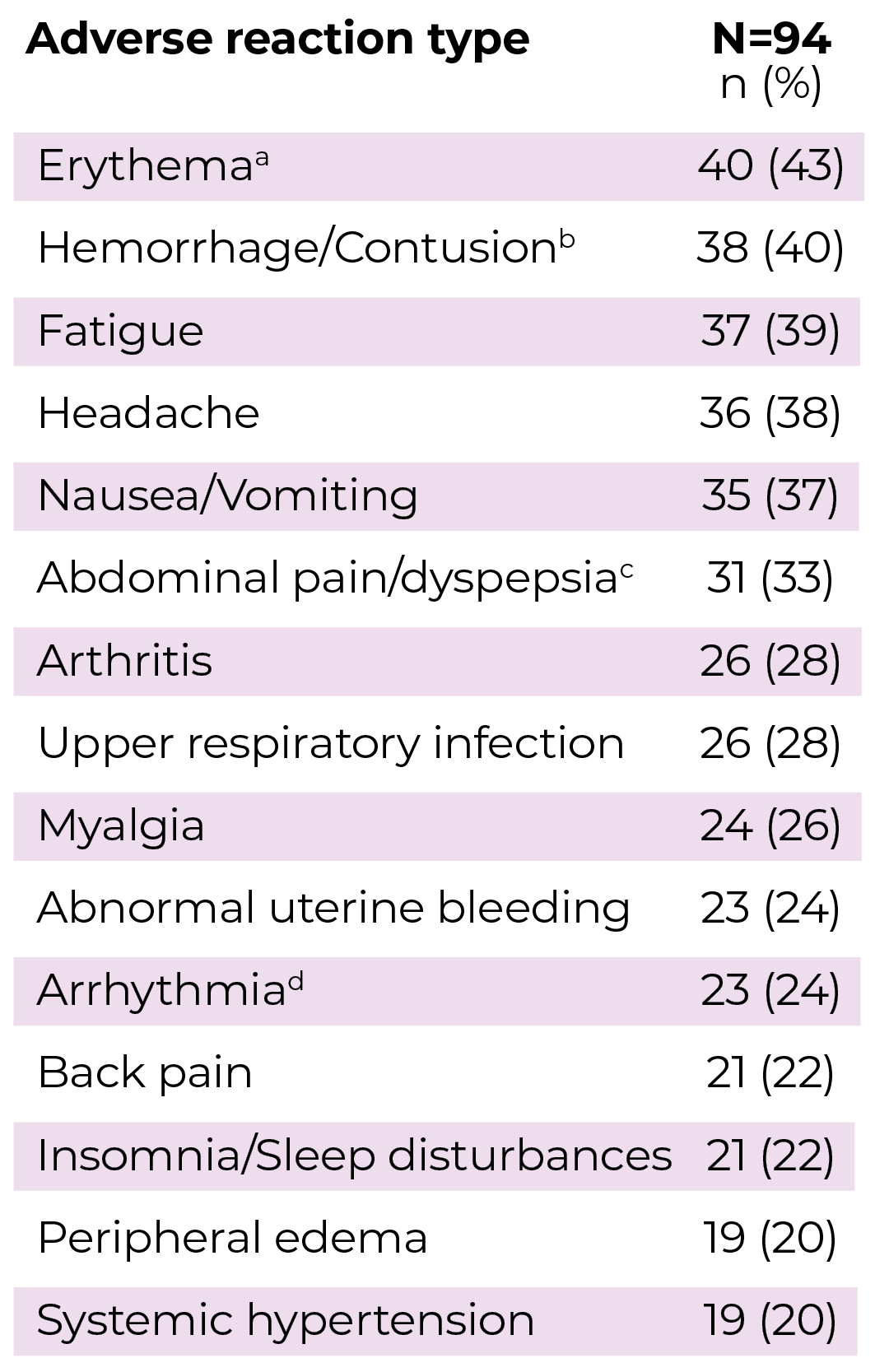
aErythema includes flushing.
bHemorrhage/Contusion includes blood urine present, conjunctival hemorrhage, ecchymosis, epistaxis, hematoma, hyphemia, and red blood cells urine.
cAbdominal pain/dyspepsia includes abdominal discomfort, abdominal distension, dyspepsia, gastritis, and other related terms.
dArrhythmia includes bradycardia, carotid pulse increased, defect conduction intraventricular, electrocardiogram QT prolonged, electrocardiogram T wave abnormal, heart rate increased, palpitations, and sinus bradycardia.
Pooled safety data (N=166) for hepatic injury and other liver-related adverse reactions and elevations in AST or ALT: at least one liver-related adverse reaction (27%), liver enzyme elevation* (20%), and AST or ALT > ULN† (45%).
*Liver enzyme elevation refers to elevation in AST, ALT, alkaline phosphatase, or gamma-glutamyl transferase.
†N=155; Not all elevations in liver enzymes were reported as adverse reactions during the studies.
ALT=alanine aminotransferase; AST=aspartate aminotransferase; TEAE=treatment-emergent adverse event; ULN=upper limit of normal.
References: 1. RECORLEV [prescribing information]. Chicago, IL; Xeris Pharmaceuticals, Inc. 2. Open-label treatment in Cushing’s Syndrome (OPTICS). ClinicalTrials.gov identifier:NCT03621280. Updated June 19, 2025. Accessed December 15, 2025. https://clinicaltrials.gov/study/NCT03621280. 3. Pivonello R, Zacharieva S, Elenkova A, et al. Levoketoconazole in the treatment of patients with endogenous Cushing’s syndrome: a double-blind, placebo-controlled, randomized withdrawal study (LOGICS). Pituitary. 2022;25(6):911-926. 4. Fleseriu M, Pivonello R, Elenkova A, et al. Efficacy and safety of levoketoconazole in the treatment of endogenous Cushing’s syndrome (SONICS): a phase 3, multicentre, open-label, single-arm trial [published correction appears in Lancet Diabetes Endocrinol. 2019;7(11):e22]. Lancet Diabetes Endocrinol. 2019;7(11):855-865. 5. Fleseriu M, Auchus RJ, Greenman Y, et al. Levoketoconazole treatment in endogenous Cushing’s syndrome: extended evaluation of clinical, biochemical, and radiologic outcomes. Eur J Endocrinol. 2022;187(6):859-871.