The SONICS study evaluated the efficacy and safety of RECORLEV® in patients with Cushing’s syndrome in a multicenter, open-label study consisting of 3 phases.
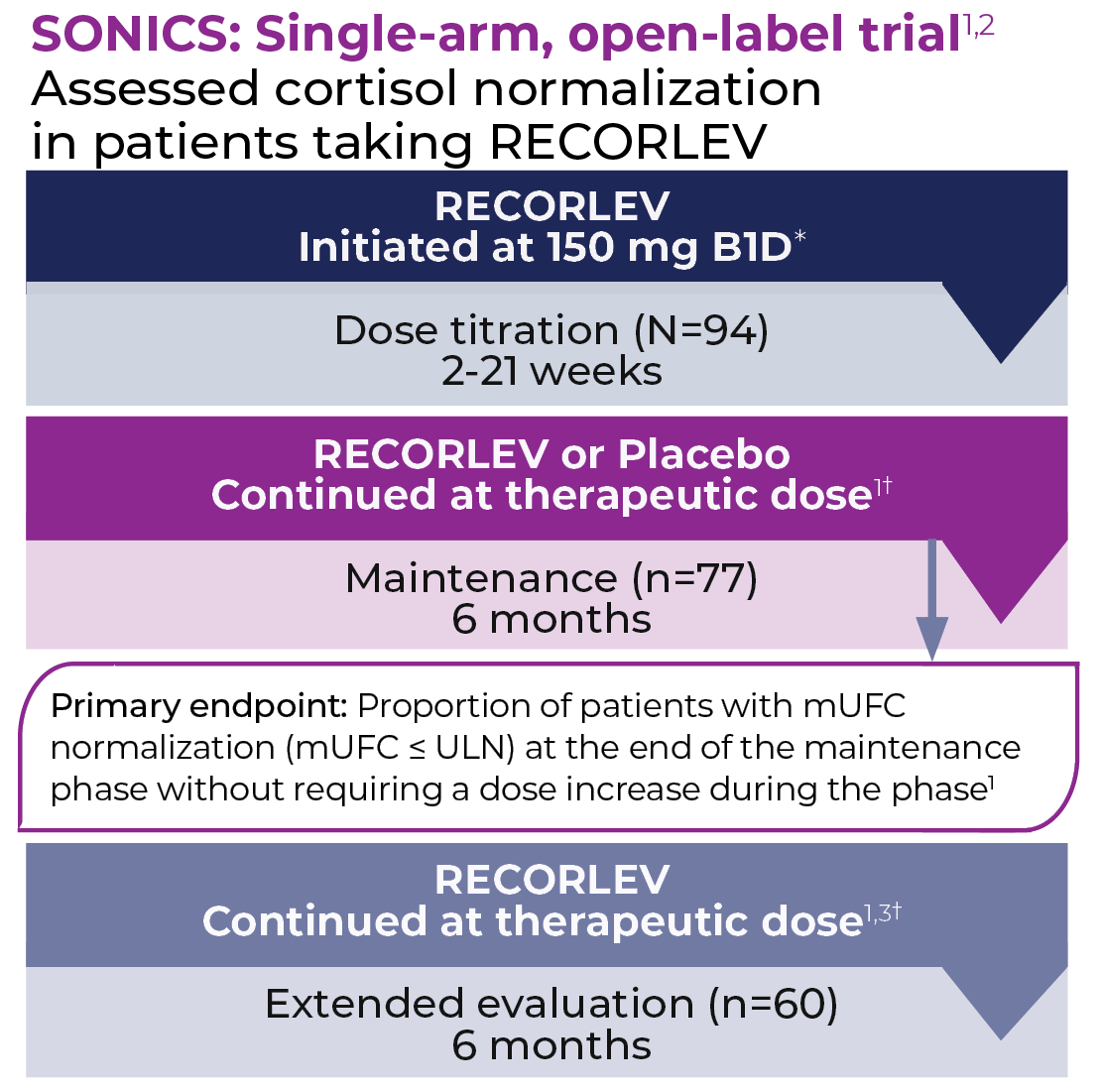

* Dose was titrated approximately every 2-3 weeks if mUFC >ULN to a maximum of 600 mg orally BID. Patients continued to the maintenance phase when they achieved a therapeutic dose.1
† Therapeutic dose was defined as a dose at which mUFC was ≤ULN or maximum allowed dose (600 mg orally BID) had been reached, or a clinically meaningful partial response based on clinical judgment, and the maximum tolerated dose had been reached. Therapeutic dose remained unchanged unless adjustments were necessary to maintain cortisol control or to address safety or tolerability issues.1
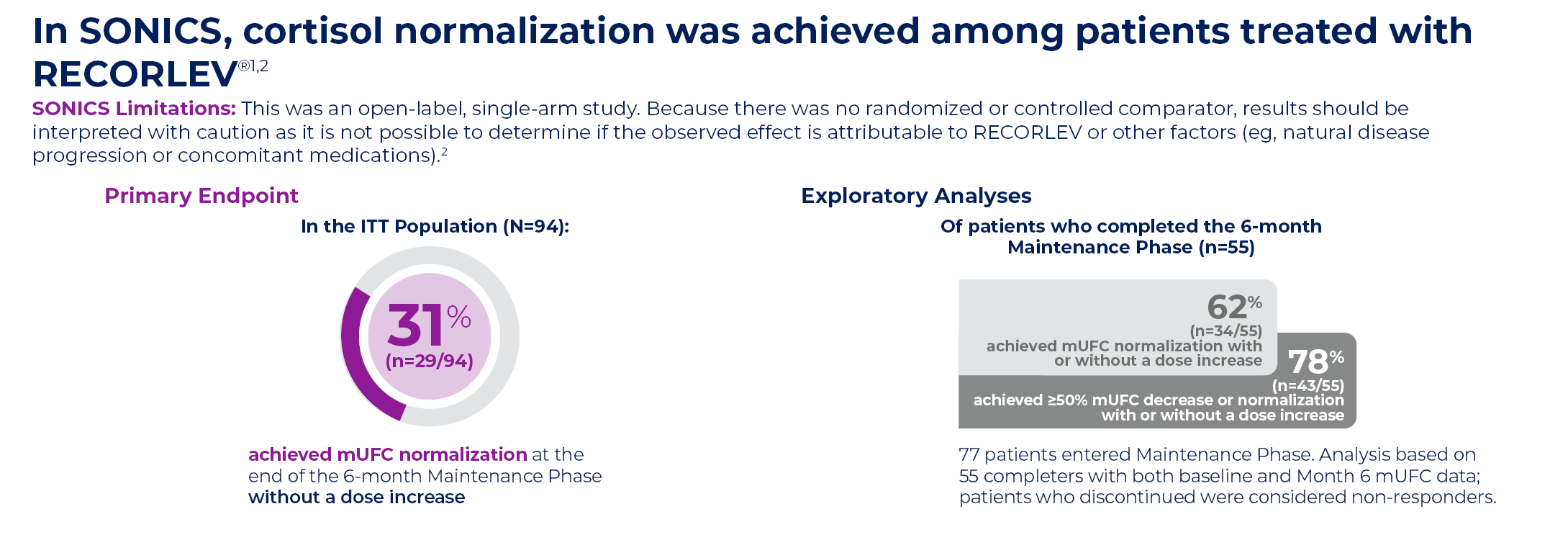

ITT, intention-to-treat; mUFC=mean urinary free cortisol; ULN=upper limit of normal.
References: 1. Recorlev [prescribing information]. Chicago, IL: Xeris Pharmaceuticals, Inc 2. Fleseriu M, Pivonello R, Elenkova A, et al. Efficacy and safety of levoketoconazole in the treatment of endogenous Cushing’s syndrome (SONICS): a phase 3, multicentre, open-label, single-arm trial [published correction appears in Lancet Diabetes Endocrinol. 2019;7(11):e22]. Lancet Diabetes Endocrinol. 2019;7(11):855-865. 3. Fleseriu M, Auchus RJ, Greenman Y. Levoketoconazole treatment in endogenous Cushing’s syndrome: extended evaluation of clinical, biochemical, and radiologic outcomes. Eur J Endocrinol. 2022;187(6):859-871.